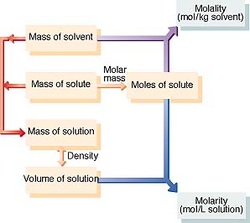
Molarity - number of moles of solute in one liter of solution
M = amount of solute (mol) / volume of solution (L)
Molality - the concentration of a solution expressed in moles of solute per kilograms of solvent
m = moles of solute / mass of solvent (kg)
A dilution solution is one that contains a relatively small amount of the solute in a given volume of solvent. Tap water is an example of a dilute solution ; it contains very small quantities of dissolved minerals. A concentrated solution contains a relatively large amount of the solute in the same volume of solvent.
M = amount of solute (mol) / volume of solution (L)
Molality - the concentration of a solution expressed in moles of solute per kilograms of solvent
m = moles of solute / mass of solvent (kg)
A dilution solution is one that contains a relatively small amount of the solute in a given volume of solvent. Tap water is an example of a dilute solution ; it contains very small quantities of dissolved minerals. A concentrated solution contains a relatively large amount of the solute in the same volume of solvent.
